Our Services
-

Transcription
Convert your audio to text.
-

Subtitles
Add captions to your videos.
Supported Languages
Below is the list of popular languages we support for transcription and subtitles.
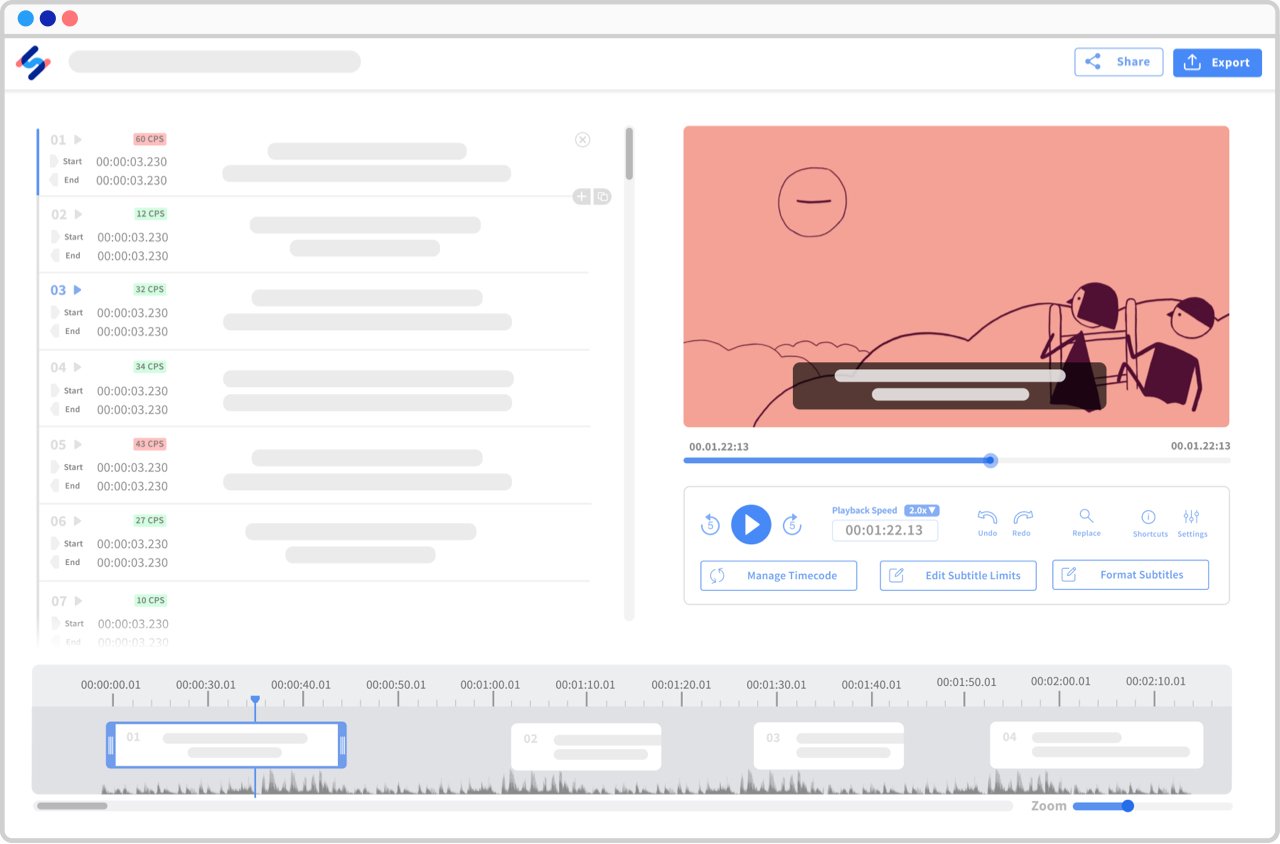
See all languages ▸Interactive editors, endless possibilities
Made for transcribers and subtitlers, our interactive editors will ease the way you interact with your transcripts and subtitles.
View a sample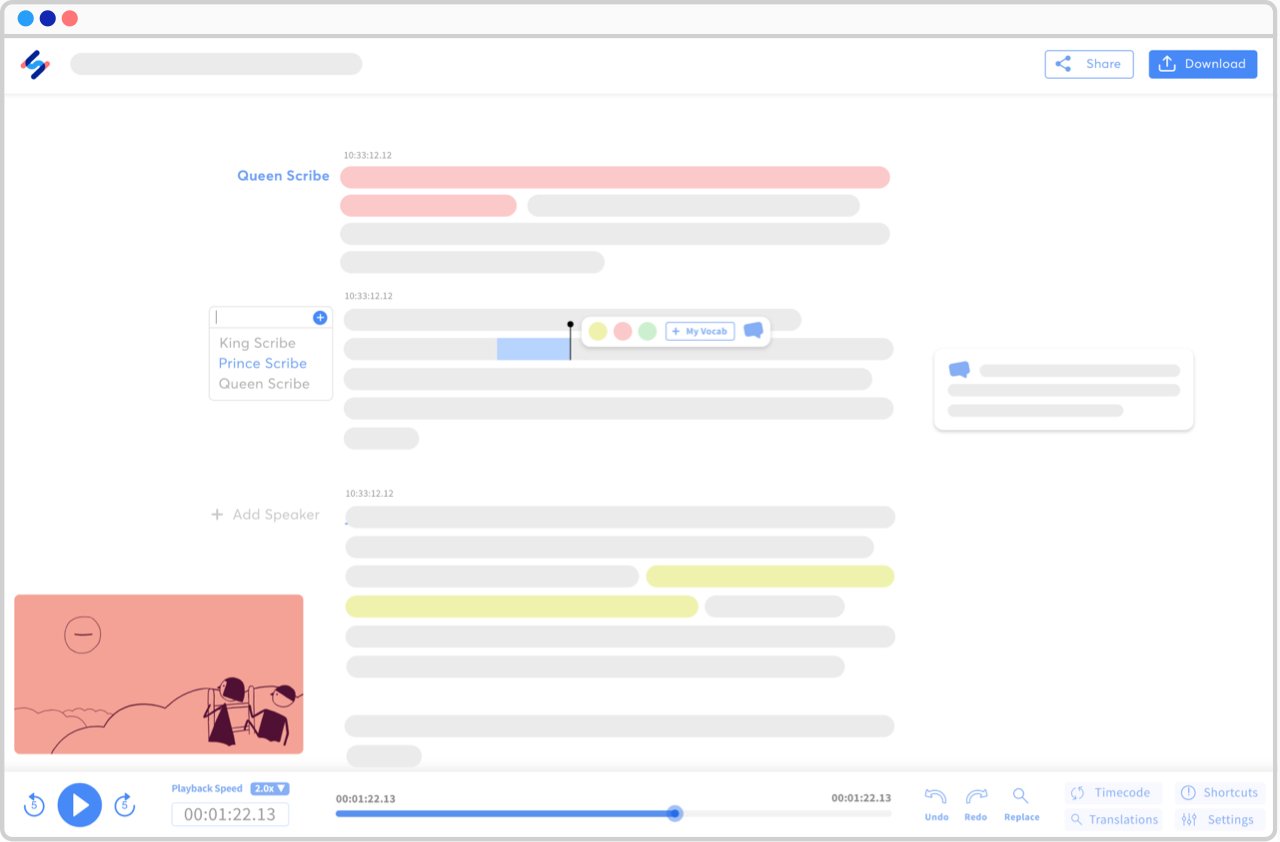
-
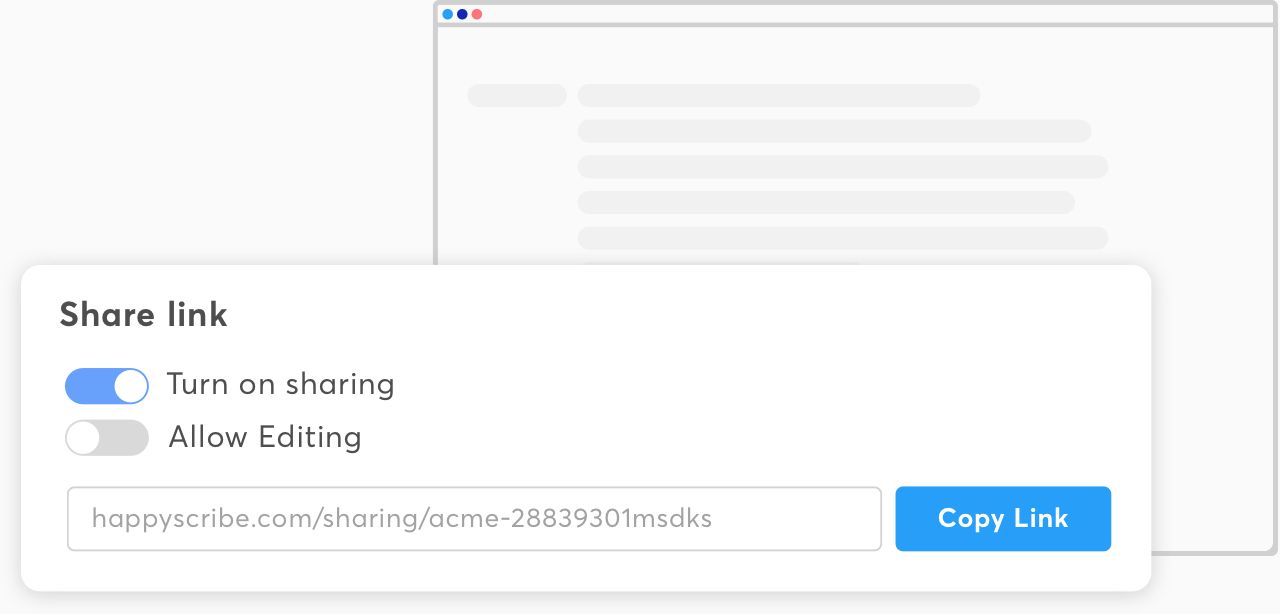
Sharing in a snap
Collaborate with all your stakeholders by sharing your transcripts and subtitles in view-only or edit mode — no matter where they are in the world.
-
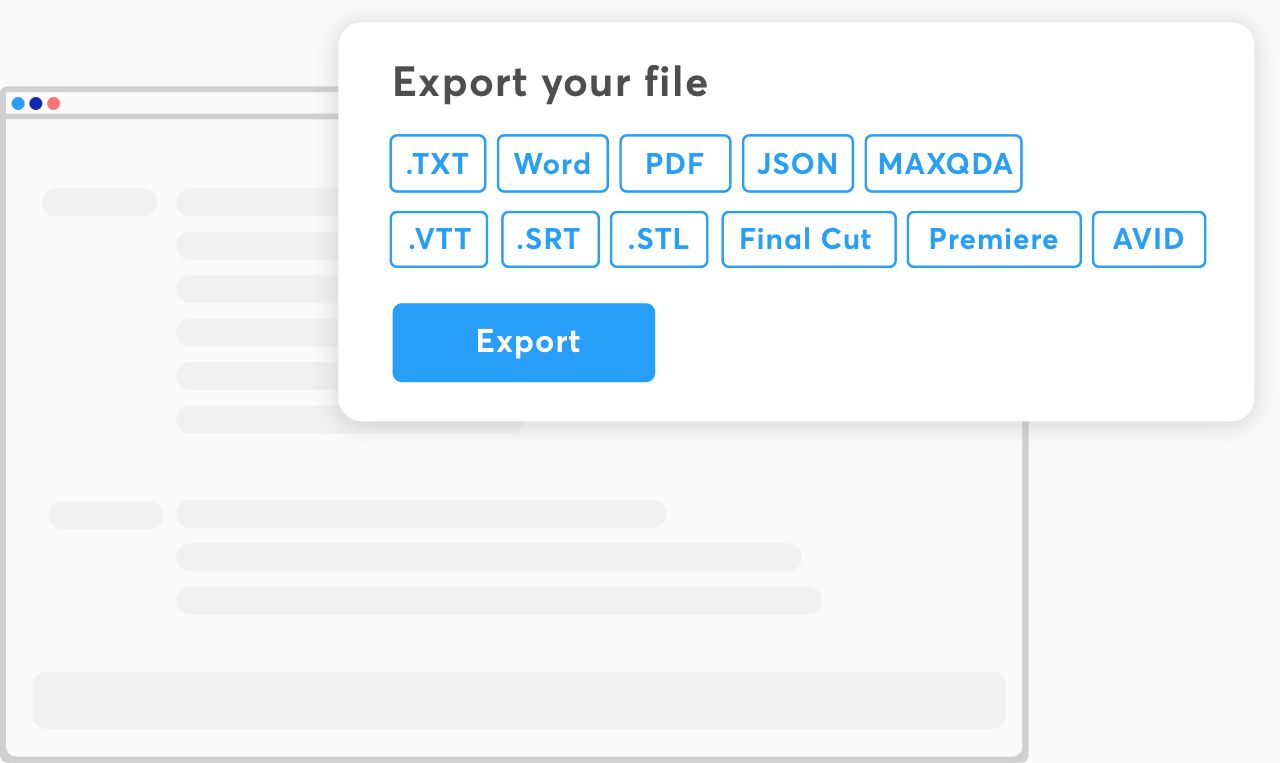
Multiple export formats
Export in all formats that you can think of. Our platform prepares you files that are ready for any kind of platform.
-
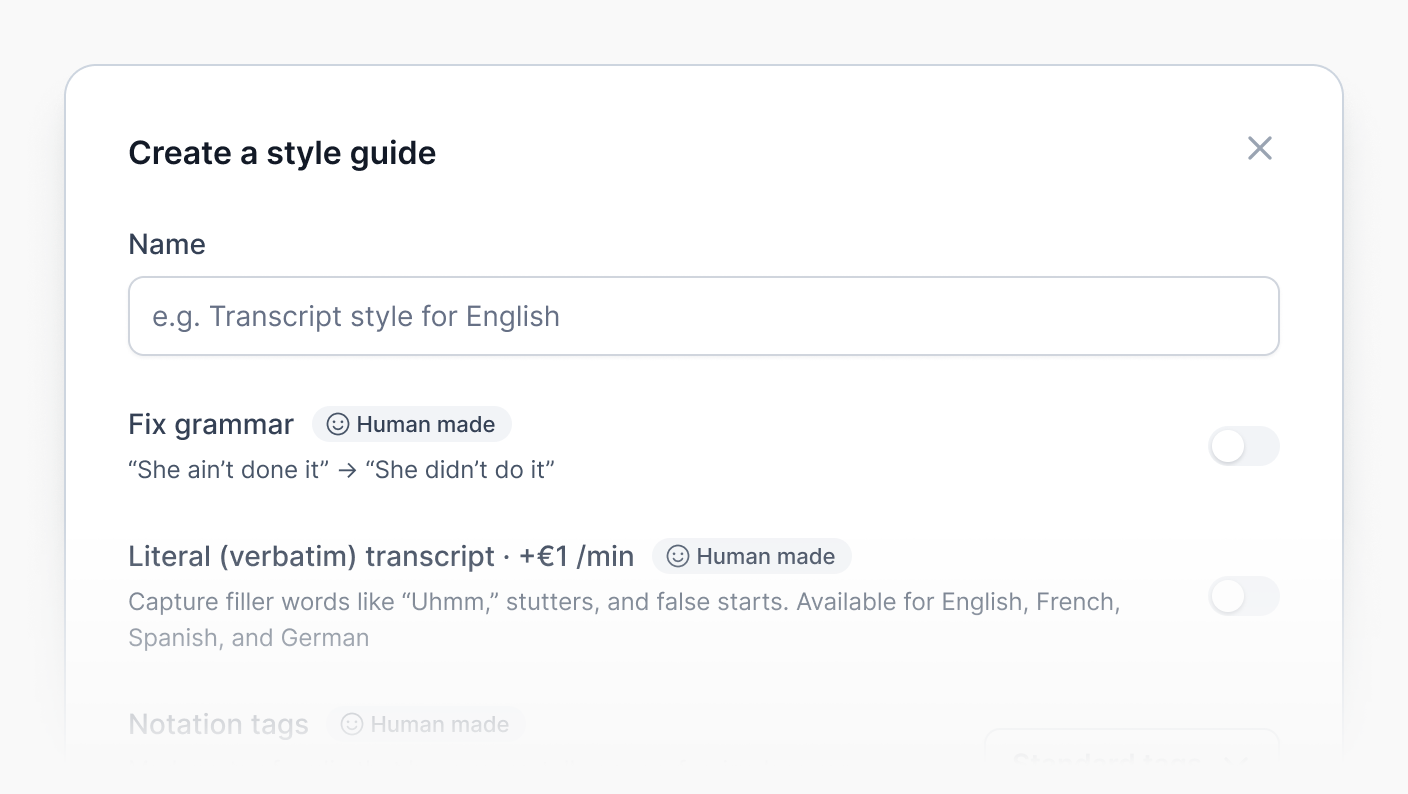
Netflix-quality subtitles with our style guide
Our style guide is tailored for project managers who demand precision, customization, and efficiency for their subtitling projects: choose your settings for timing, formatting, advanced grammar corrections, speaker identification, and more.
-
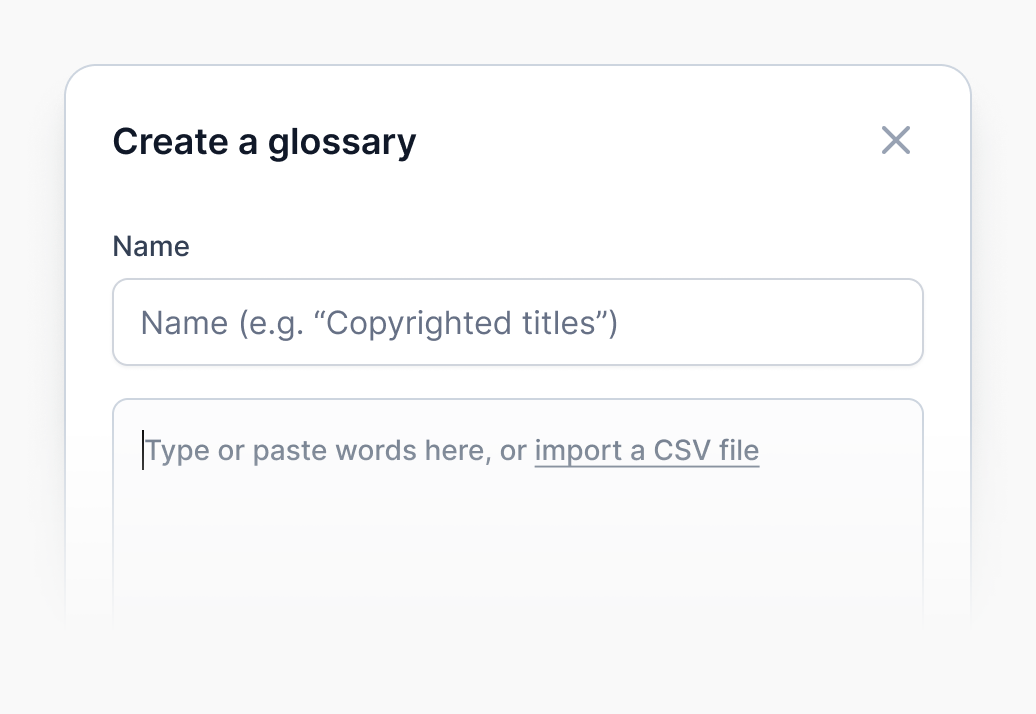
Glossaries and special guidelines
By adding specialized terminology (brands, slang, etc.) to a glossary, you can significantly reduce review time. We also support special guidelines, including SDH and verbatim, alongside a dedicated team of professional linguists.
Features
-
No limits on uploads
Upload files of any size and length. Our software supports them all.
-
Machine Translation
Automatically translate your transcription and subtitles in the most common languages.
-
Import from other platforms
Import any public links and synchronize Happy Scribe to your current workflow.
-
Workspaces for collaboration
Create spaces for you to share your files with the rest of your team.
-
APIs & Integrations
Seamlessly integrate with your favorite applications: Zapier, Youtube, and more.
-
Security & Confidentiality
All files are protected and remain private. Your subtitles are protected.
-
Perfect for transcribingThis website is perfect for transcribing any video fast and with ease. To make it even better, it has numerous useful features like translation, etc. I highly recommend it.Yahia Mostafa - Trustpilot
-
The product and team are fantasticThe product and team are fantastic. Can't think of anything negative about the company.Everard Brockett - Trustpilot
-
Awesome way to combine subtitle (SRT)…Awesome way to combine subtitle (SRT) files and Video - especially for youtube music videosJoshua - Trustpilot
-
It met my needsIt met my needs. The program struggles with homophones and proper names, but otherwise is very accurate.Landmark Inn Staff - Trustpilot


